Carbon atom1/11/2024 Carbon is made in the interiors of stars, although it was not produced in the Big Bang.Elemental carbon can take the form of one of the hardest substances (diamond) or one of the softest (graphite).Because it forms more compounds than any other element, it is sometimes called the "King of the Elements." Carbon is a nonmetal that can bond with itself and many other chemical elements, forming over ten million compounds.Many other common organics also include oxygen, nitrogen, phosphorus, and sulfur. The simplest organic molecules consist of carbon chemically bonded to hydrogen. Carbon is the basis for organic chemistry, as it occurs in all living organisms.Similar to this, amides are also classified as primary, secondary and tertiary based on the number of carbons connected to the nitrogen. These are called quaternary ammonium salts. There is a little difference in the way amines are classified! Unlike the previous cases, the amines are classified based on the number of carbons connected to the nitrogen:Īnother difference with the amines is that the nitrogen can have four groups connected by using the lone pair and getting a positive formal charge. 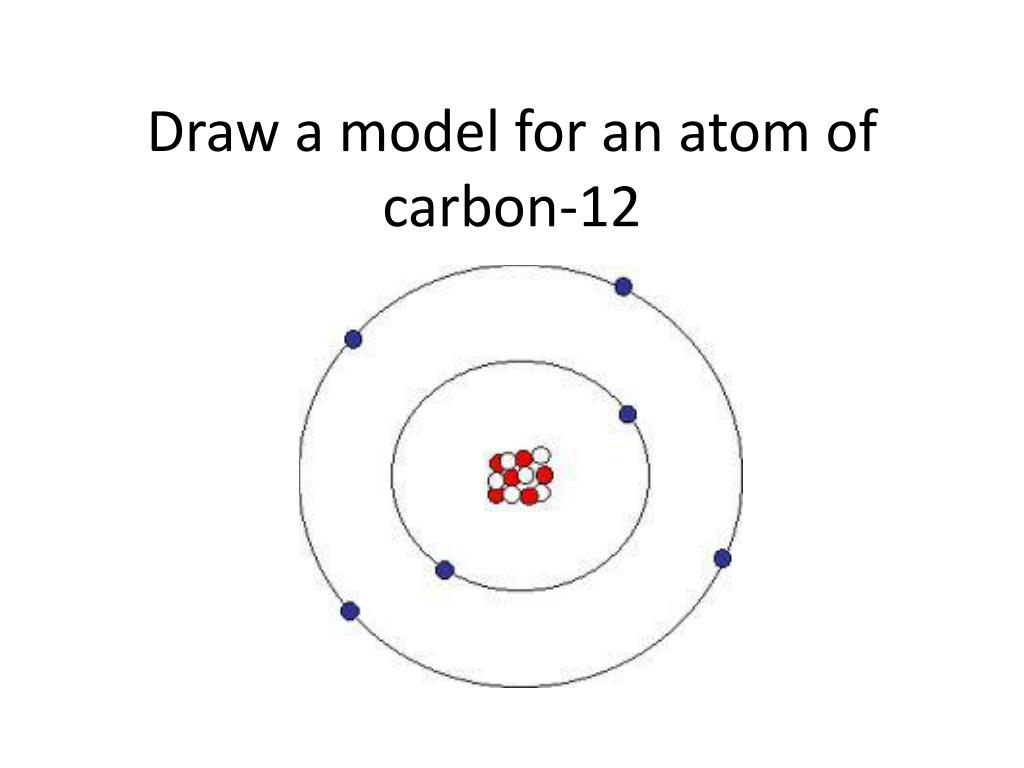
To distinguish between a primary, secondary or a tertiary alkyl halide, locate the carbon that is connected to the halogen and count how many carbon atoms are connected to it:ĭepending on the number of carbon atoms connected to the one with the hydroxyl group, the alcohols are also classified as primary, secondary and tertiary: This classification will be especially important in the nucleophilic substitution and elimination reactions. Primary, Secondary and Tertiary Alkyl Halides There is no such thing as a quaternary carbocation since the four connected carbons satisfy the octet and the central carbon cannot have a formal charge. You need to count how many carbon atoms are connected to the positively charged carbon. The principle for classifying the carbon atoms is the same. The carbocations range from methyl carbocation to tertiary carbocation. Primary, Secondary and Tertiary Carbocations This designation of carbon atoms is also applied to carbocations, alkyl halides, alcohols, amines and amides. Depending on which carbon of the butyl is connected to the parent chain, or another group, we have an n-butyl, sec-butyl, and tert-butyl.Īnd these are simply the abbreviations for the carbon’s degree of unsaturation: secondary- sec, tertiary- tert. For example, butane has four isomers and therefore, four isomeric alkyl groups. 
The central carbon is connected to three carbon atoms of the CH 3 groups:įinally, if four carbon atoms are connected to a carbon then we have a quaternary carbon:īefore moving on to other functional groups, let’s see how the degree of substitution (1 o, 2 o, 3 o) is also used to distinguish certain fragments in naming organic molecules. The following isomer of butane contains a tertiary carbon. However, the middle carbon (CH 2) is connected to two carbon atoms, therefore it is a secondary carbon. The CH 3 carbons are primary as they are only connected to the CH 2 carbon. Starting from propane, we can locate a primary and a secondary carbon. Notice that it is a symmetrical molecule and regardless which carbon you choose, it is going to be a primary carbon, sine there is going to be only one carbon connected to it. We can mark this carbon in red and circle the ones connected to it in blue. And for this, you need to count the number of carbons that are directly connected to the carbon of interest. The carbons in ethane can, however, be classified. The first molecule in this series is methane and because there is only one carbon, it is not classified by any of the definitions we just discussed. You will encounter this notation when starting to learn the functional groups and continue using it in the nomenclature of organic compounds and later in the semester. Tertiary carbons are connected to three carbon atoms.Īnd if four carbons are connected to a carbon, then it is a quaternary carbon. Secondary carbons are connected to two carbon atoms. Primary carbons are connected to one carbon only. In short, these definitions are assigned to carbon atoms based on the number of other carbon atoms they are connected to: 
This is also referred to as the carbon degree of substitution. That is the definition of primary, secondary, tertiary and quaternary carbon atoms in organic chemistry. Today, we will talk about a terminology that you are going to use very often in your organic chemistry 1 class.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
